FDA Issues First COVID-19 Vaccine EUA for Pfizer-BioNTech, December 11, 2020

Date: December 11, 2020


Date: December 11, 2020
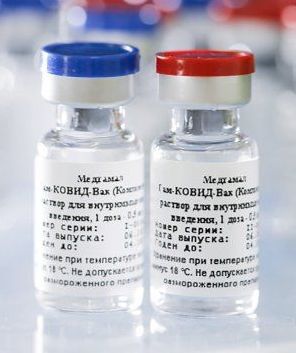
Russia begins production on the Sputnik V COVID-19 vaccine. Date: August 15, 2020 Read more on Wikipedia → 📅 Other events on August 15 📅 Events on this day in 2020 📅 On This Day (Today) More events from 20202020 U.S. Presidential Election: Biden vs. Trump, November 3, 2020Second Nagorno-Karabakh War Armistice Agreement Signed by…
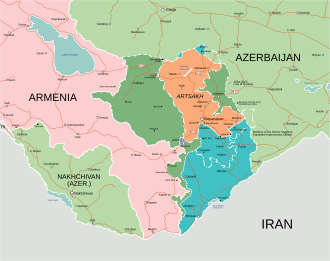
Second Nagorno-Karabakh War: Azerbaijan launches an offensive against the self-proclaimed Republic of Artsakh in the Nagorno-Karabakh region, inhabited predominantly by ethnic Armenians. Date: September 27, 2020 Read more on Wikipedia → 📅 Other events on September 27 📅 Events on this day in 2020 📅 On This Day (Today) More events from 2020FDA Issues First…

Indian Prime Minister Narendra Modi announces the country's largest ever self-imposed curfew, in an effort to fight the spread of COVID-19. Date: March 22, 2020 Read more on Wikipedia → 📅 Other events on March 22 📅 Events on this day in 2020 📅 On This Day (Today) More events from 2020China National Day of…

An oil spill in Norilsk releases 17,500 tons of diesel oil into nearby rivers. Date: May 29, 2020 Read more on Wikipedia → 📅 Other events on May 29 📅 Events on this day in 2020 📅 On This Day (Today) More events from 2020Trump Acquitted by Senate in First Impeachment Trial, February 5, 2020Trump…
A grand jury in Kentucky declines to indict three police officers for the shooting death of Breonna Taylor in a drug raid gone wrong, leading to nationwide protests in the U.S. Date: September 23, 2020 Read more on Wikipedia → 📅 Other events on September 23 📅 Events on this day in 2020 📅 On…

Lewis Hamilton wins the Turkish Grand Prix and secures his seventh drivers' title, equalling the all-time record held by Michael Schumacher. Date: November 15, 2020 Read more on Wikipedia → 📅 Other events on November 15 📅 Events on this day in 2020 📅 On This Day (Today) More events from 2020Vega Rocket Fails After…